Two TCR Discovery Routes
Covering the Complete pHLA-TCR Solution Space
Route A: Antigen-driven (pHLA→TCR) | Route B: TCR-sequencing-library-driven (TCR→pHLA)
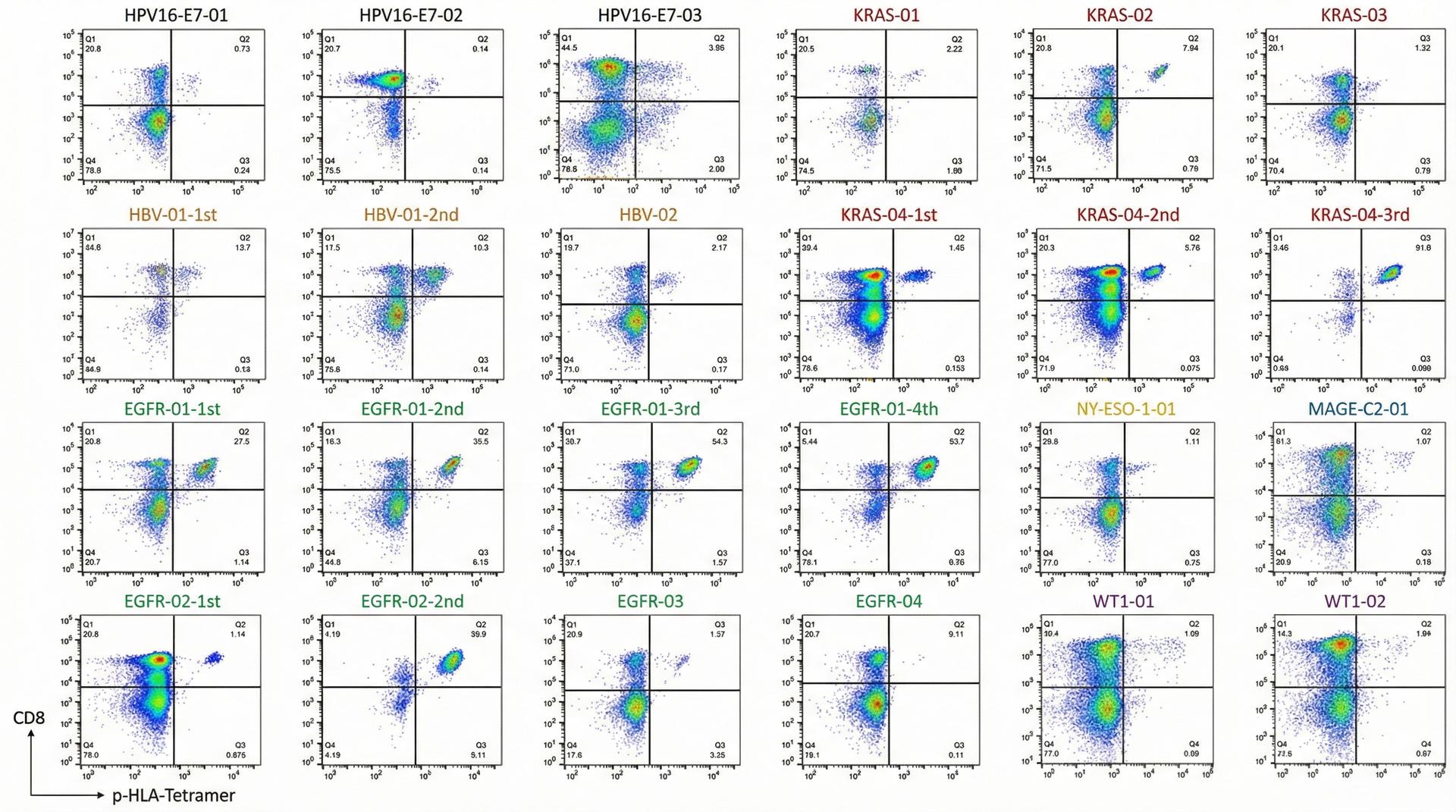
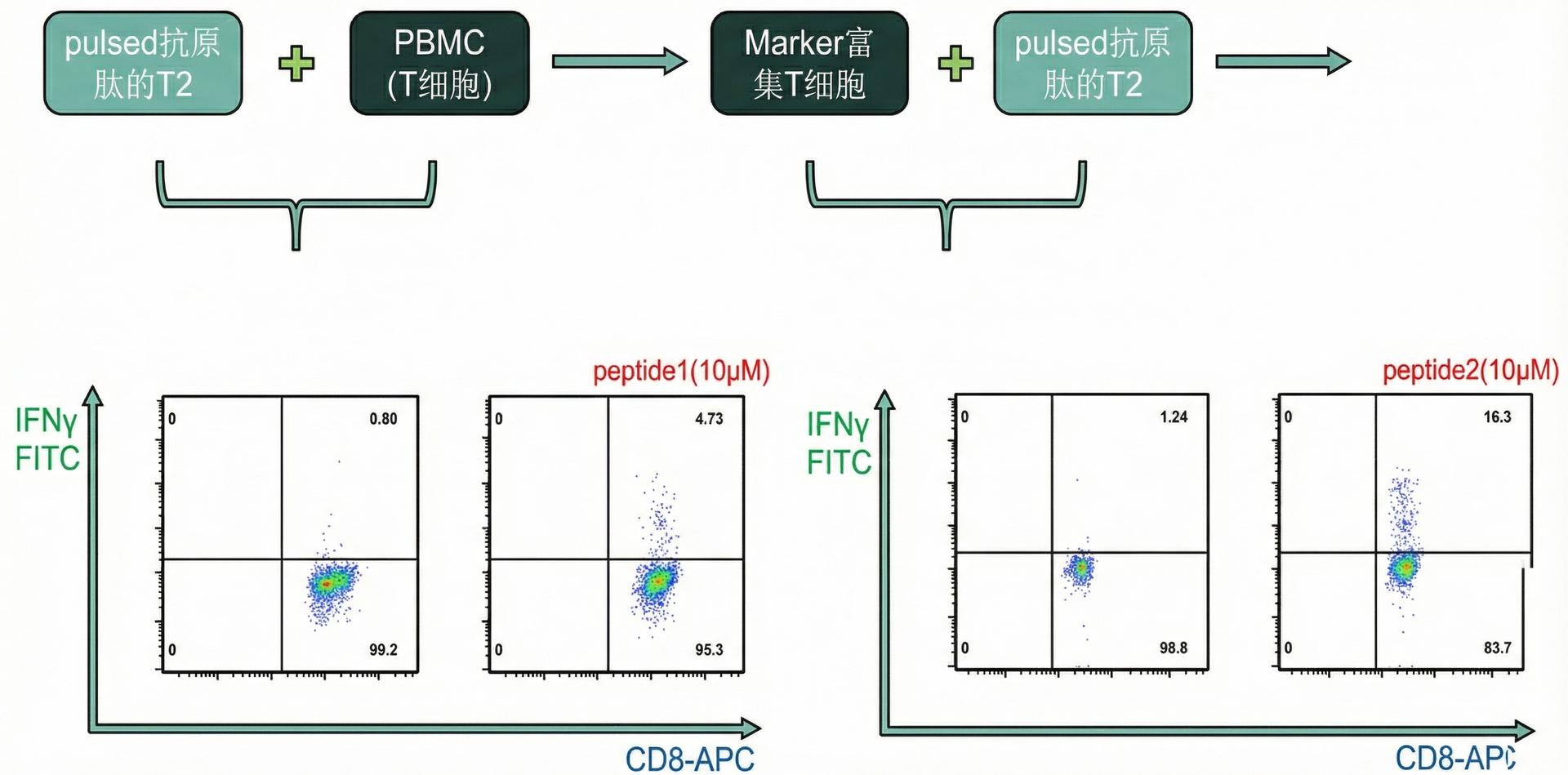
INFINITY-TCR operates two complementary TCR discovery routes. Route A (antigen-driven), based on our proprietary Allogeneic-HLA TCR Priming, delivers 30-40× efficiency, completing CTL discovery in as few as 45 days — our main recommendation. Route B (reverse screening) serves clients with existing TCR sequencing data but no antigen information. 3 patents published/filed, 8 hospital collaborations.
vs. Conventional Methods
Discovery Cycle
Published / Filed
Hospitals · Research Institutes